Thank you to all our partners and sponsors for their support and contributions to our research activities.
Un immense merci à nos collaborateurs et sponsors pour leur soutien.

Thank you to all our partners and sponsors for their support and contributions to our research activities.
Un immense merci à nos collaborateurs et sponsors pour leur soutien.

Virtual reality for surgical training

Robot-aided percutaneous nephrolithotomy
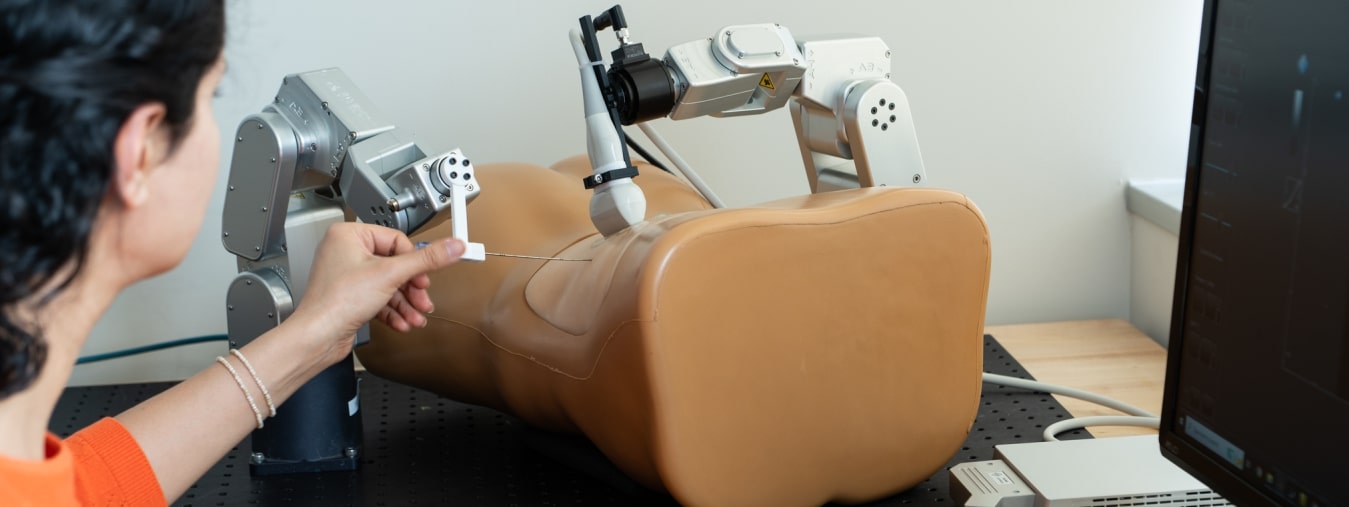
Wideband dielectric spectroscopy for tissue biopsy
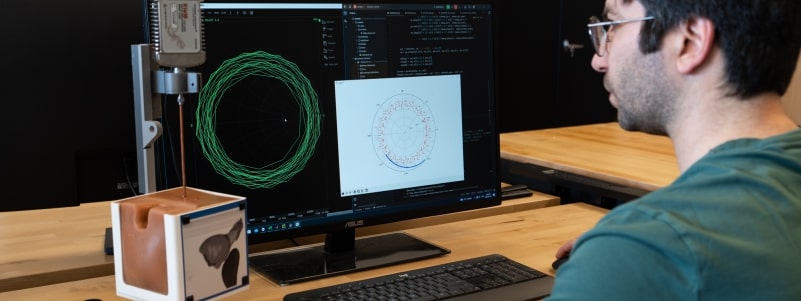
Ultrasound image simulation for surgical training

Acoustoelectric tomography for targeted percutaneous surgery

Haptics and haptic devices for robotic surgery

AI for pediatric laparoscopy training

 Smart firefighter helmet using simultaneous localization and mapping
Smart firefighter helmet using simultaneous localization and mapping

Structural firefighting is undoubtedly one of the most dangerous professions. At times, firefighters inside unknown structures can become disoriented due to heavy smoke and a potentially changing environment in the event of a ceiling or wall collapse.
This is an extremely dangerous scenario and can be fatal if the firefighter is not able to find their way out or be rescued. In partnership with the City of Oshawa and Oshawa Fire Services, we have developed
a smart helmet (shown on the left) that can build a dynamic map of the smoky environment as a firefighter enters a structure. The map, similar to a floorplan, is then used to localize the firefighters within the structure and send
information about their location to an Incident Commander (IC). This allows the IC to know the exact location of each firefighter inside a structure at all times, even in previously unknown environments. In the event of a Firefighter
mayday, since the exact location of the firefighter is now known, rescue measures can be optimized in order to bring that Firefighter back to safety as quickly as possible.
Researcher(s): Sam Lovett & Tyler Paquette (2020), Karl Kanmaz & Brayden DeBoon (2019).
 Actuators for robotic-assisted rehabilitation
Actuators for robotic-assisted rehabilitation
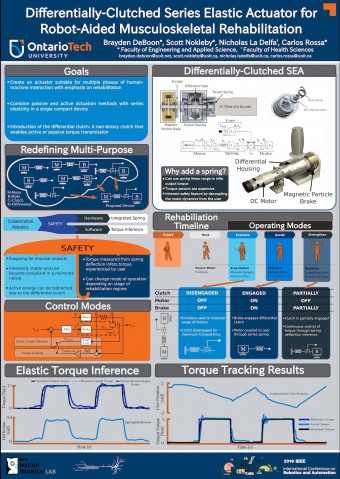
Musculoskeletal disorders (MSDs) are the leading cause of lost-time injury and worker compensation costs in Canada, placing a significant burden on individuals, workplaces, and the health care system. Telemedicine is a prominent vehicle to provide healthcare
to patients at a distance and give access to medical services that would otherwise not be available in distant communities. However, the current MSD telemedicine practice is mostly within the audio-visual domain, which significantly
limits the practice of occupational and physical therapy.
We are developing remotely-operated devices with the ability to translate the motion of a therapist at the patient’s home and enable MSD rehabilitation
to be conducted through existing telemedicine networks across the country. The actuator on the left was developed as a means of bridging this gap by translating the therapists motion to the patient in a safe and effective manor. The
goal is to provide meaningful assistance to people experiencing MSDs who are limited in either mobility or access to appropriate medical facilities.
Researcher: Brayden DeBoon.
 Hybrid actuators for haptic devices
Hybrid actuators for haptic devices

Traditionally, haptic interfaces use active actuators such as electric motors to generate force feedback. It is well-known that these interfaces can suffer from instability issues depending on the simulated environment. In several applications, instability
is a particular concern as it represents a true danger to the user. Passive actuators such as brakes and dampers are intrinsically stable and safe. We demonstrated that small-scale magnetorheological (MR) fluid brakes combined with
small DC motors can increase the performance and safety of force-feedback devices. The picture on the left shows the hybrid actuator we developed comprising two unidirectional MR brakes and a DC motor. The complete system can produce
7.9 times more torque than a volume-equivalent DC motor with lower inertial and frictional torques. The device has been patented and transferred to one of our industry partners in Europe.
Read the news story published on eeNews Europe: here.
 Robot-assisted brachytherapy
Robot-assisted brachytherapy

One out of eight Canadian men will be diagnosed with prostate cancer during their lifetime. In Canada, prostate cancer accounts for roughly one-quarter of all cancer cases in men, making it the leading cause of cancer deaths with 24,000 diagnoses and
4,100 deaths every year. Brachytherapy is a popular treatment for men with early prostate cancer due to its high success rate, minimal side effects, and patient convenience. It involves the insertion of needles loaded with tiny radioactive
seeds into the prostate. Once the needles are fully inserted, they are pulled back to permanently leave the seeds in locations inside the prostate, where the radiation released from them treats the cancer cells. The radioactive seeds
must be implanted in target locations with great accuracy. However, current techniques only enable surgeons to place seeds to within 5 mm of an intended target.
The figure on the left shows the hand-held instrument we developed that helps surgeons place seeds with an accuracy of 0.33 mm. It steers the needle automatically by rotating its base as the surgeon inserts it. Delivering
radiation with pinpoint accuracy will lead to better prostate cancer control and fewer side effects for patients. It can also allow treating prostate cancer situations that are not possible with the current imprecise technique. One
example is treating only a part of the prostate for early prostate cancers.
 High performance magnetorheological brakes for haptic devices
High performance magnetorheological brakes for haptic devices
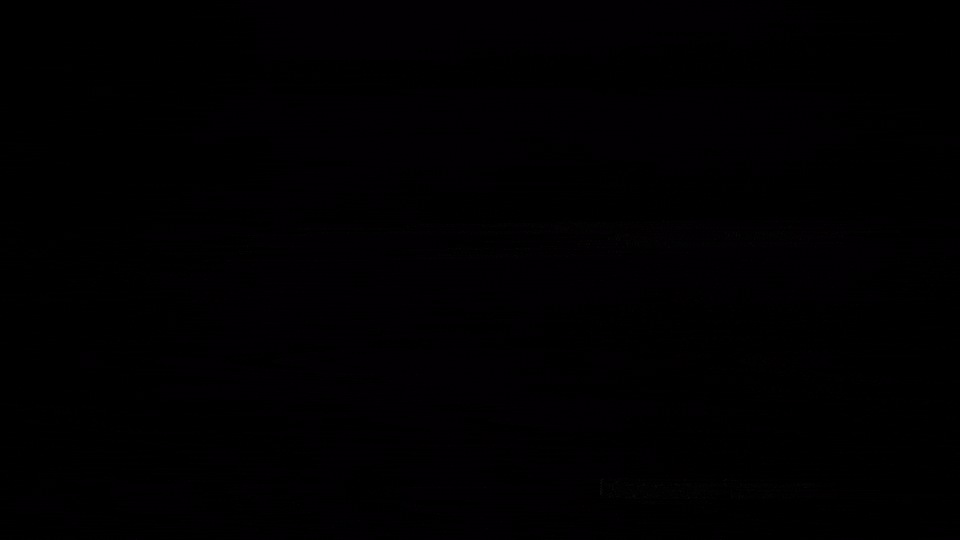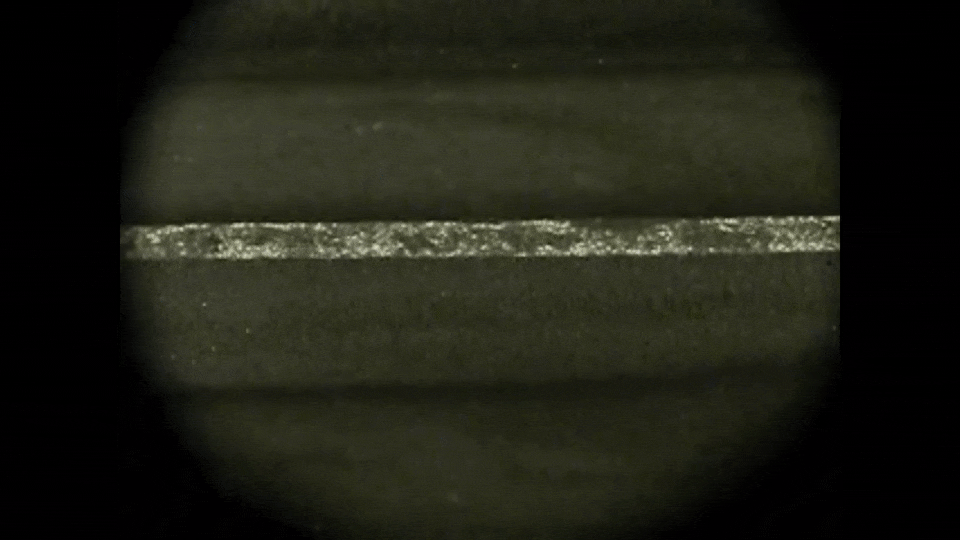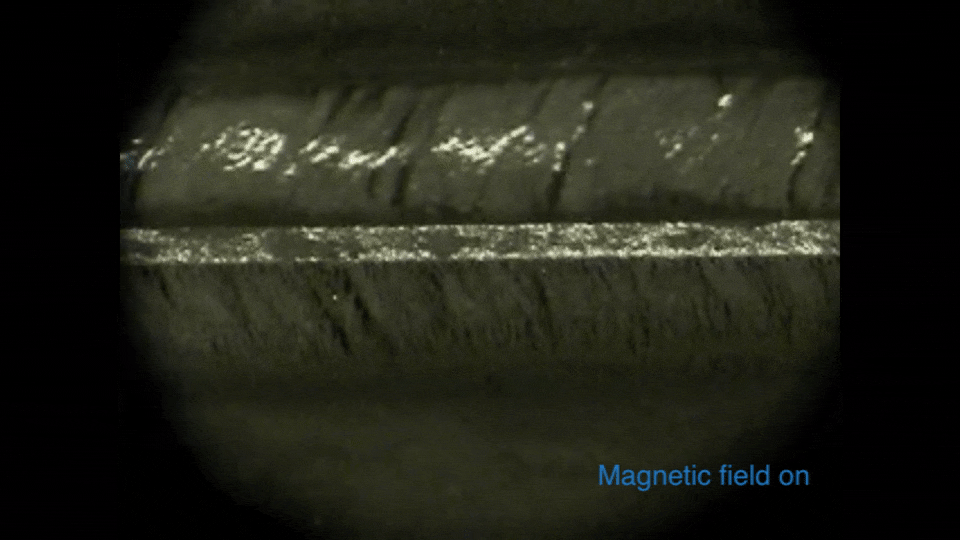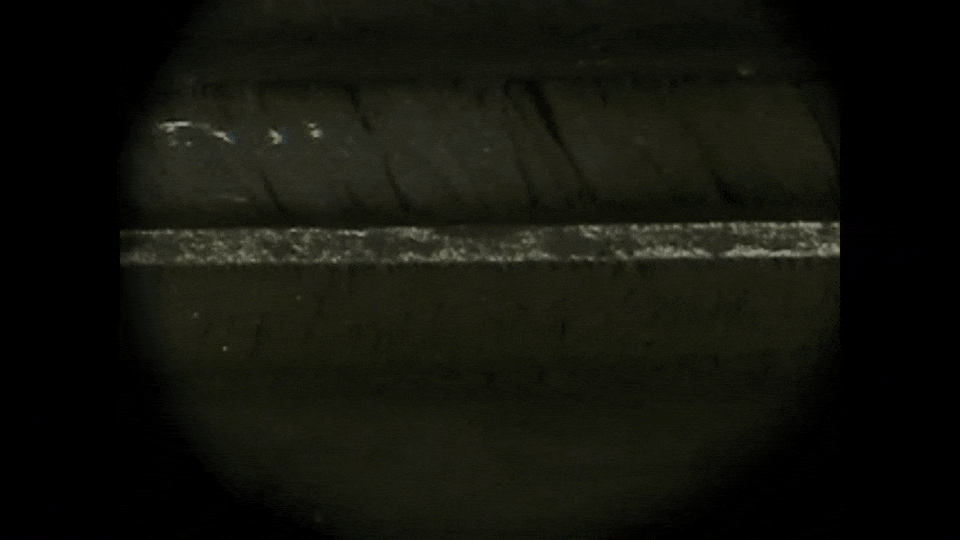
Magnetorheological (MR) fluids have emerged as a promising technology for new actuator design. An MR fluid is an active material composed of a suspension of soft ferromagnetic micron-sized particles (typically 1 to 10 microns) dispersed in a carrying
liquid (mineral oils, synthetic oils or water). Under the action of an external magnetic field, these particles form chain-like structures or aggregates aligned roughly parallel to the magnetic field, changing the apparent viscosity
of the fluid (see the animation on the left).
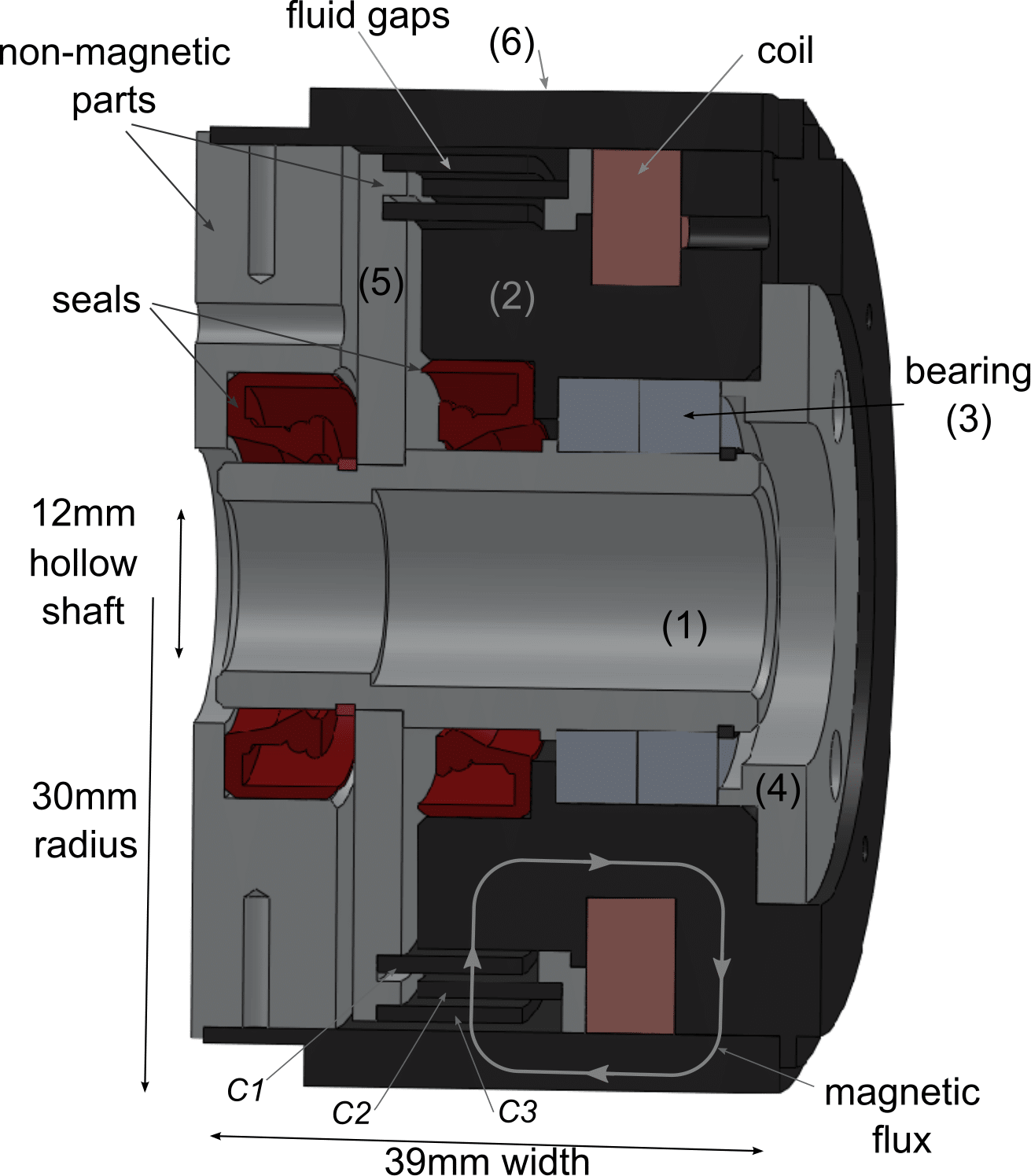
Classical approaches for designing MR brakes have not accounted for fluid nonlinearity and magnetic saturation while addressing tradeoffs in terms of repose time, off-state torque, power supply, desired torque and volume. We showed
that performance is strongly dependent on electromechanical design and introduced a new model for optimal actuator design. This resulted in an actuator with 23% more torque in a volume 76% smaller than a commercially available MR brake
(second figure).